Corrosion in steel products is a significant issue, impacting industries worldwide. Understanding "what causes steel products to corrode" is crucial for maintaining structural integrity. According to recent studies by the Corrosion Prevention Association, over 30% of annual maintenance costs in industrial settings stem from corrosion issues. This highlights the economic burden it places on businesses.
Dr. Emily Johnson, a leading corrosion expert, states, "Corrosion is not just a material degradation; it is an environmental challenge that requires our attention." Various factors contribute to the deterioration of steel, including moisture, salt exposure, and chemical reactions. Each scenario presents unique risks and potential costs.
With this in mind, recognizing the top causes of corrosion can help mitigate these challenges. Curious stakeholders must appreciate that ignoring corrosion leads to catastrophic failures. Decision-makers should invest in research and preventative measures. The consequences of neglecting this issue are often dire and costly.
Corrosion is a natural process that deteriorates metals. It occurs when metal reacts with moisture, air, or various chemicals. Steel, in particular, is highly susceptible to corrosion. Understanding corrosion types is crucial for preventing damage. Two common types are uniform corrosion and pitting corrosion. Uniform corrosion affects the entire surface. Pitting causes localized damage and is harder to detect.
According to a study by the National Association of Corrosion Engineers, corrosion costs the U.S. economy over $276 billion annually. This represents about 3.1% of the Gross Domestic Product. Another report highlights that about 30% of steel in construction experiences some form of corrosion. With such significant impact, industry professionals must prioritize effective prevention strategies.
Environmental factors greatly influence steel corrosion. This includes humidity, salt exposure, and pollutants. For example, coastal areas see higher rates of corrosion due to saltwater. Additionally, certain industrial processes can accelerate corrosion rates. Regular inspections and maintenance can save costs in the long run. However, many organizations overlook these vital steps, resulting in avoidable failures.
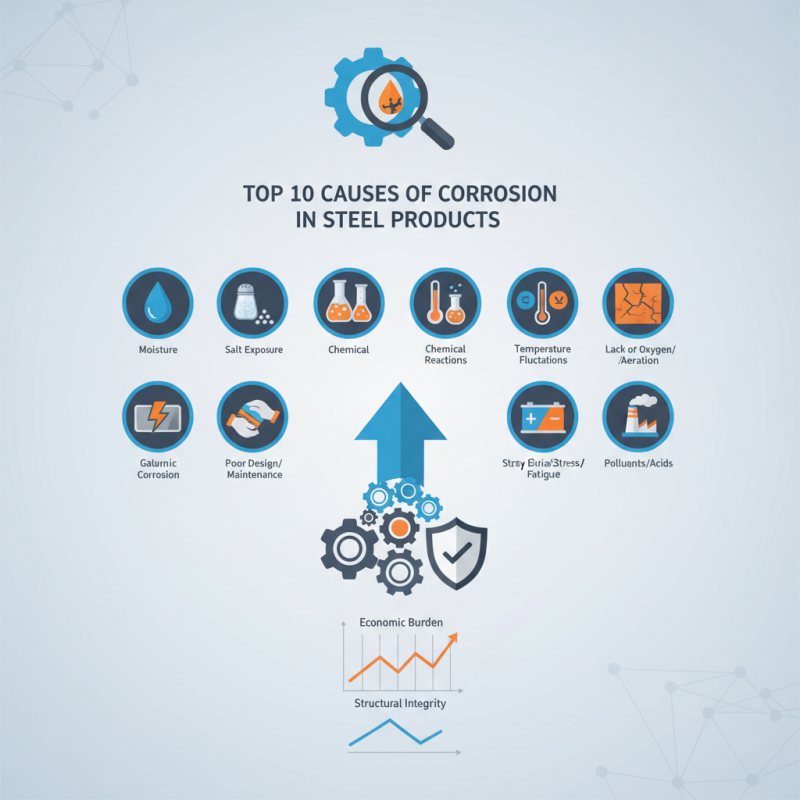
This bar chart represents the top 10 causes of corrosion in steel products, demonstrating the most prevalent factors affecting material integrity.
Corrosion in steel products is influenced by various environmental factors. One major factor is moisture. High humidity levels can accelerate the rusting process. Steel surfaces attract water, creating an environment where corrosion thrives. This is especially common in coastal areas. Here, the salt in the air further exacerbates the problem.
Another significant factor is temperature. Extreme temperature fluctuations can cause steel to expand and contract. This motion can create cracks and openings, allowing corrosive agents to penetrate. In industrial settings, exposure to chemicals can also lead to rapid deterioration. It's important to assess the types of substances nearby.
Tips: To prevent corrosion, ensure proper ventilation in storage areas. This reduces humidity and moisture buildup. Applying protective coatings can also create a barrier against environmental factors. Regular inspections help identify early signs of rust. Small cracks can lead to bigger issues later.
Ignoring these factors may result in costly repairs. Reflections on maintenance routines can help spot weaknesses. Implementing proactive measures can mitigate these risks. The environment plays a crucial role in steel integrity, so staying informed is essential.
Corrosion in steel products often stems from various chemical reactions. One common cause is the presence of moisture. When water interacts with iron in steel, it triggers oxidation. This process produces iron oxide, commonly known as rust. Rust forms a reddish-brown coating, leading to structural weaknesses over time.
Another factor is acidic environments. Acids can react aggressively with steel, accelerating the corrosion process. For instance, exposure to hydrochloric acid can significantly weaken steel. Even rainwater, slightly acidic due to dissolved carbon dioxide, can contribute to corrosion over time. The relentless cycle of wet and dry conditions can exacerbate these reactions.
Lastly, environmental pollutants can accelerate corrosion. Sulfur dioxide and nitrogen oxides may fall as acid rain, increasing penetration into the steel. It's crucial to consider how these reactions can interact with each other. Even small amounts of these elements can lead to significant damage. Understanding these chemical processes might guide better protective strategies. Addressing galvanic corrosion is equally vital. Contact between different metals can initiate additional corrosion pathways. Identifying these risks is essential to combat steel degradation effectively.
| Cause of Corrosion | Description | Chemical Reaction Type | Preventive Measures |
|---|---|---|---|
| Rusting | Oxidation of iron in the presence of moisture. | Oxidation-Reduction | Use protective coatings, maintain dry conditions. |
| Galvanic Corrosion | Occurs when two dissimilar metals are in contact. | Electrochemical | Insulate metals from each other, use sacrificial anodes. |
| Pitting Corrosion | Localized corrosion forming small pits or holes. | Electrochemical | Use corrosion-resistant alloys, apply protective coatings. |
| Crevice Corrosion | Occurs in stagnant environments in small gaps. | Electrochemical | Eliminate crevices, ensure good drainage. |
| Uniform Corrosion | Even loss of material across the surface. | Electrochemical | Regular maintenance, use of corrosion inhibitors. |
| Stress Corrosion Cracking | Cracking due to tensile stress and corrosive environment. | Electrochemical | Control stress levels, use stress-relief techniques. |
| Microbiologically Influenced Corrosion (MIC) | Caused by the presence of microorganisms. | Biochemical | Maintain hygiene, use biocides where necessary. |
| High Temperature Corrosion | Accelerated corrosion at elevated temperatures. | Oxidation | Use heat-resistant alloys, control operating temperature. |
| Environmental Corrosion | Corrosion due to exposure to environmental conditions. | Varied mechanisms | Protective barriers, regular inspections. |
Corrosion in steel products is a significant concern. However, preventive measures can greatly reduce its impact. Keeping steel dry is vital. Moisture accelerates corrosion. Regular inspections make it easier to catch early signs. Even the smallest rust spots should be addressed quickly.
Applying protective coatings can also help. Paints and sealants act as barriers against moisture and chemicals. In some cases, galvanization provides extra protection. Choosing the right coating is essential for effectiveness. Regular maintenance can extend the lifespan of steel products.
Environmental factors should not be overlooked. Exposure to salt, humidity, and air can increase risk. Using corrosion inhibitors can offer additional defense. An imperfect understanding of these factors often leads to issues. Companies may neglect proper training on corrosion prevention. Awareness and proactive strategies can make a crucial difference.
Regular maintenance is crucial in reducing corrosion risks in steel products. Corrosion can weaken structures, leading to expensive repairs. Failing to check for signs of corrosion regularly can have long-term consequences. A proactive approach helps in early detection and prevention.
Tips for maintenance include the following. Inspect steel surfaces frequently for rust or peeling paint. Address these issues immediately. Keep steel surfaces clean by washing off dirt and contaminants. Moisture can lead to corrosion. Ensure your environment is dry to reduce risk.
Inadequate maintenance can cause serious problems. Neglecting protective coatings leaves steel vulnerable. Corroded areas may spread quickly if not managed. It's essential to stay vigilant and make repairs when necessary. Consciously investing time in maintenance pays off in the long run.
: Improved convenience and efficiency in daily tasks. Users often notice significant time savings.
Yes, many users find it user-friendly. However, some may need time to adapt to its features.
Absolutely, it offers versatility. Users can use it for various tasks based on their needs.
Durability varies by usage. Regular maintenance can extend its lifespan. Some users suggest checking periodically.
Yes, occasional technical glitches reported. Users may need to troubleshoot or seek support if issues arise.
Regular cleaning enhances performance. Follow the guidelines for proper care to avoid damage.
Understand the limitations. Some users feel overwhelmed initially. Take time to learn its functionalities.
Warranties may vary. Check the terms for specifics. Some users appreciate having coverage for peace of mind.
Corrosion is a significant issue affecting steel products, primarily due to environmental and chemical factors. Understanding what causes steel products to corrode involves recognizing both the environmental conditions, such as humidity and temperature changes, and the chemical reactions that occur when steel reacts with moisture and various chemicals. These reactions can lead to rust and deterioration, severely compromising the integrity of steel structures.
Preventive measures, including protective coatings and regular maintenance, are crucial in mitigating corrosion risks. Maintaining steel products through inspections and timely repairs not only prolongs their lifespan but also ensures safety and performance in various applications. By being aware of the primary causes of corrosion and implementing effective strategies, industries can significantly reduce the impact of this pervasive problem on steel products.

 Campfire Steel
Campfire Steel